Objectives : to be able to measure the initial rate of enzyme activity.
Equipment :
Method :
Dilute the Trypsin enzyme using distilled water
Put different concentrations of Trypsin; 0.2%,0.4%,0.6%, and 0.8%, in the test tubes, using different ratios to get the correct concentration.
Then add the milk powder suspension into the cuvettes along with the trypsin concentration.
Immediately place the cuvette into the colorimeter and start the stop watch.
Measure the light transmittance every 15 seconds until 100% of the light is transmitted through the solution.
Repeat these steps with all the concentrations of trypsin.
Equipment :
- Milk powder suspension
- Trypsin solution (1%)
- 6 test tubes and test tube rack
- Stop watch
- Pippettes (5cm3)
- Safety glasses
- Colorimeter
- 5 cuvettes
- Distilled water
Method :
Dilute the Trypsin enzyme using distilled water
Put different concentrations of Trypsin; 0.2%,0.4%,0.6%, and 0.8%, in the test tubes, using different ratios to get the correct concentration.
Then add the milk powder suspension into the cuvettes along with the trypsin concentration.
Immediately place the cuvette into the colorimeter and start the stop watch.
Measure the light transmittance every 15 seconds until 100% of the light is transmitted through the solution.
Repeat these steps with all the concentrations of trypsin.
Conclusion of the experiment :
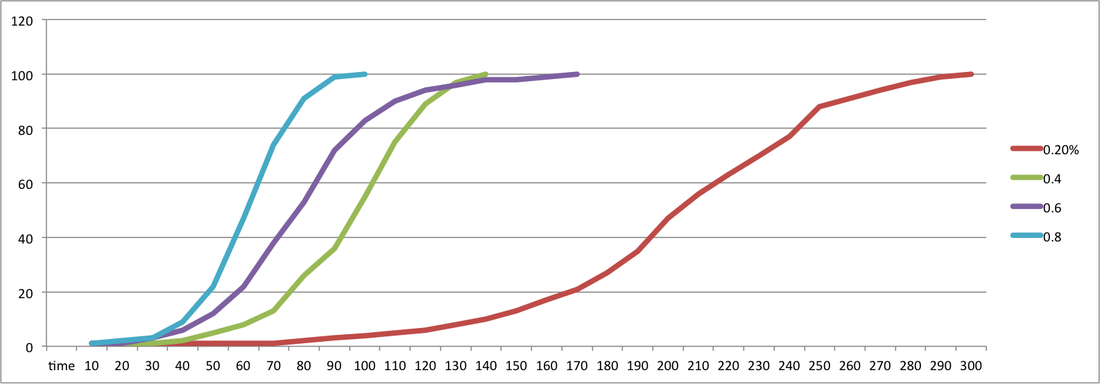
In conclusion the results of my experiment show that the higher concentration of the enzyme Trypsin the faster the digestion of the milk powder suspension. This is shown in my results by the 0.2% concentration of Trypsin digesting the milk powder suspension in 300 seconds which is the slowest of all the concentrations. This continues with the other concentrations, the 0.4% concentration reaction finishes after 140 seconds, the 0.6% concentration reaction however takes slightly longer which doesn't conform with the trend. Although the 0.8% concentration did follow the trend and the reaction was the shortest time at 100 seconds.
The faster reaction time for the higher concentration is due to there being more enzyme particles in the higher concentration and less milk particles therefore they would react faster and the reaction would be shorter
In conclusion the results of my experiment show that the higher concentration of the enzyme Trypsin the faster the digestion of the milk powder suspension. This is shown in my results by the 0.2% concentration of Trypsin digesting the milk powder suspension in 300 seconds which is the slowest of all the concentrations. This continues with the other concentrations, the 0.4% concentration reaction finishes after 140 seconds, the 0.6% concentration reaction however takes slightly longer which doesn't conform with the trend. Although the 0.8% concentration did follow the trend and the reaction was the shortest time at 100 seconds.
The faster reaction time for the higher concentration is due to there being more enzyme particles in the higher concentration and less milk particles therefore they would react faster and the reaction would be shorter
Questions :
- IV- the concentration of the trypsin. DV- the initial rate of reaction.
- The initial rate of reaction is important to use because the first few seconds is when the results vary the most and when the results are the easiest to compare, if an average rate of reaction was used then the time towards the end of the experiment would cause the results to become more unclear and less precise.
- If the cuvette is scratched it would cause random errors because the cuvette used for the reaction would be scratched whereas the reference cuvette wouldn't be scratched. It isn’t systematic because not all the results would be inaccurate as not all the cuvettes were scratched.
- Temperature is one variable that would normally be controlled in enzyme catalyst reactions, it could be controlled by using a water bath to control the temperature of the solutions, this is important because each enzyme has an optimum temperature that if exceeded can cause the enzyme to denature. Ph is another variable that would normally be controlled in enzyme catalyst reactions, it could be controlled by using Ph probes to monitor the Ph and then add acid or alkali to alter the Ph if needed, this is important because each enzyme has an optimum Ph that can alter the results if the Ph varies from each test tube.
 RSS Feed
RSS Feed
