Method:
1. Draw a table for results showing date, time, hours of incubation and absorbance.
2. Using aseptic technique to prevent contamination, add 0.025 g dried yeast to 100 cm3 sterile distilled water at 30°C. Shake gently to ensure that the cells are evenly distributed and suspended.
3. Using aseptic technique, dilute 100 times by adding 1 cm3 to 99 cm3 sterile broth in a flask. This should give a starting concentration of 0.0025 g/l for your growth curve.
4. Using sterile medium as the reference, calibrate the colorimeter (i.e. set it to zero).
Note: keep this reference medium (the blank) in the refrigerator throughout experiment.
5. Shake the flask containing the yeast culture gently to distribute the cells evenly. Using aseptic technique, withdraw a 1 cm3 sample and add to 9cm3 of water.
6. Measure the absorbance of the sample you have just withdrawn. Record date, time and absorbance in the table.
7. Incubate at 30°C in a water bath.
8. Repeat Instructions 5 - 8 three times per day for the next three days (early morning, lunch-time and late afternoon if possible). If it is not possible to measure the absorbance at the time of taking the sample, place it in a sterile container, label with initials, date and time and refrigerate until convenient to do so, preferably within 24 hours. Make sure that the yeast is fully suspended before reading the absorbance.
1. Draw a table for results showing date, time, hours of incubation and absorbance.
2. Using aseptic technique to prevent contamination, add 0.025 g dried yeast to 100 cm3 sterile distilled water at 30°C. Shake gently to ensure that the cells are evenly distributed and suspended.
3. Using aseptic technique, dilute 100 times by adding 1 cm3 to 99 cm3 sterile broth in a flask. This should give a starting concentration of 0.0025 g/l for your growth curve.
4. Using sterile medium as the reference, calibrate the colorimeter (i.e. set it to zero).
Note: keep this reference medium (the blank) in the refrigerator throughout experiment.
5. Shake the flask containing the yeast culture gently to distribute the cells evenly. Using aseptic technique, withdraw a 1 cm3 sample and add to 9cm3 of water.
6. Measure the absorbance of the sample you have just withdrawn. Record date, time and absorbance in the table.
7. Incubate at 30°C in a water bath.
8. Repeat Instructions 5 - 8 three times per day for the next three days (early morning, lunch-time and late afternoon if possible). If it is not possible to measure the absorbance at the time of taking the sample, place it in a sterile container, label with initials, date and time and refrigerate until convenient to do so, preferably within 24 hours. Make sure that the yeast is fully suspended before reading the absorbance.
Aseptic technique:
1. Wash hands and sterilize work area using ethanol to kill any bacteria on the surface.
2. Light the bunsen burner, this creates an updraft to prevent bacteria landing in the broth.
3. Remove the bung from the test tube containing the broth and flame the neck. (without putting the bung down to prevent contamination)
4. stir the broth gently and remove 1cm3 and flame the neck of the test tube again before replacing the bung.
6. place the broth mixture into 9cm3 of water.
7.flame the inoculation loop again and replace the bung.
Safety:
1. make sure the ethanol is not left near the bunsen burner.
2. wear suitable eye protection.
1. Wash hands and sterilize work area using ethanol to kill any bacteria on the surface.
2. Light the bunsen burner, this creates an updraft to prevent bacteria landing in the broth.
3. Remove the bung from the test tube containing the broth and flame the neck. (without putting the bung down to prevent contamination)
4. stir the broth gently and remove 1cm3 and flame the neck of the test tube again before replacing the bung.
6. place the broth mixture into 9cm3 of water.
7.flame the inoculation loop again and replace the bung.
Safety:
1. make sure the ethanol is not left near the bunsen burner.
2. wear suitable eye protection.
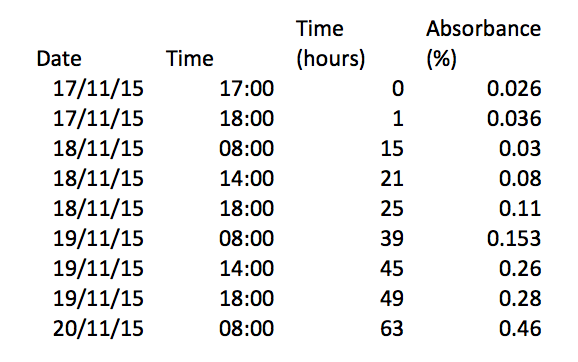
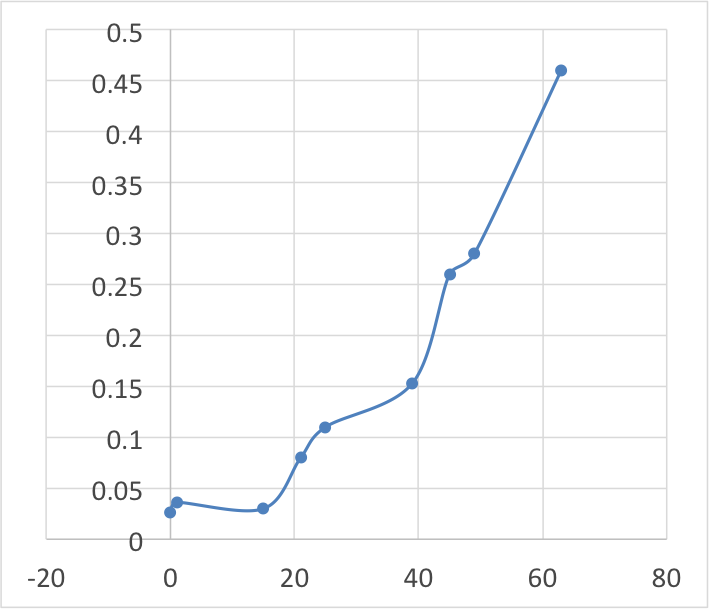
My results:
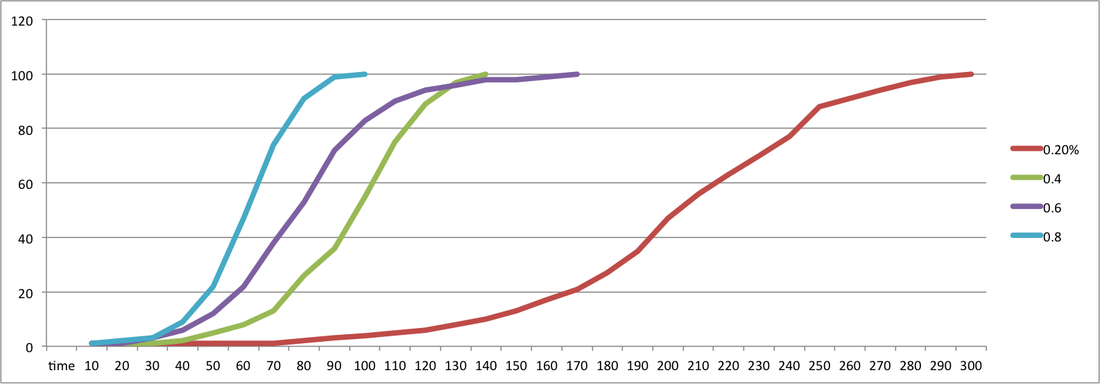
I think my results went well, some of the results were incorrect due to the change in what the colorimeter was measuring, absorbance of light/ transmission of light, we solved this by inverting the results from negative to positive. My graph shows a general upward growth that conforms with the normal bacterial growth curves lag phase and log phase. some results may also be slightly incorrect because some of the results were recorded before the broth was shaken so some sediment may have been stuck in the bottom of the conical flask.












 RSS Feed
RSS Feed
