Objective:
Know how to carry out an investigation to determine the osmotic potential and therefore water potential of plant epidermal cells.
IV-Concentration of salt solution. mol dm-3
DV-No. of cells that are plasmolysed.
Safety:
Take care with glassware, mounting needles and cutting equipment.
Equipment:
-Red onion skin with a single cell layer
-Six salt solutions of suitable range, 0.1, 0.2, 0.3, 0.4, 0.5, 0.6 mol dm-3
-Distilled water
-Six watch glasses
-Six 5cm3 syringes
-Pipettes
-Scalpel
-Tweesers
-Filter paper
-Pen
-Microscope slides and cover slips
Method:
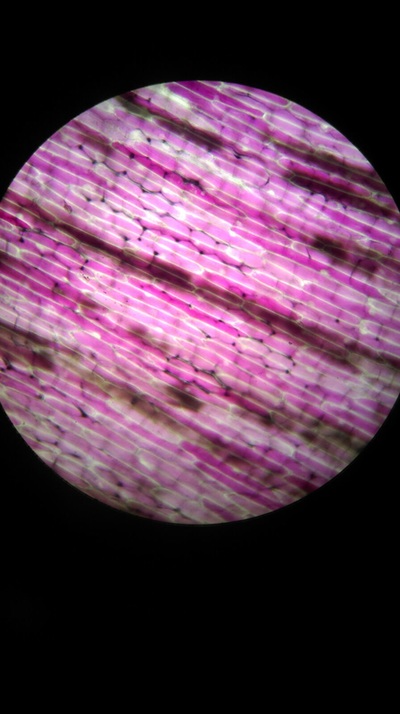
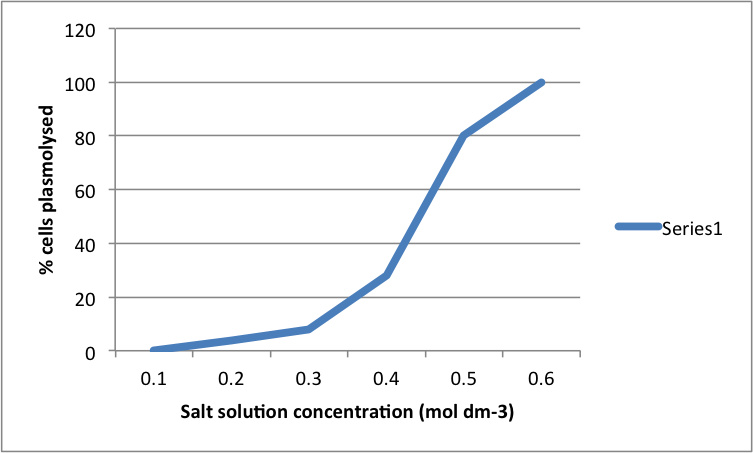
Results:
Know how to carry out an investigation to determine the osmotic potential and therefore water potential of plant epidermal cells.
IV-Concentration of salt solution. mol dm-3
DV-No. of cells that are plasmolysed.
Safety:
Take care with glassware, mounting needles and cutting equipment.
Equipment:
-Red onion skin with a single cell layer
-Six salt solutions of suitable range, 0.1, 0.2, 0.3, 0.4, 0.5, 0.6 mol dm-3
-Distilled water
-Six watch glasses
-Six 5cm3 syringes
-Pipettes
-Scalpel
-Tweesers
-Filter paper
-Pen
-Microscope slides and cover slips
Method:
- Take 6 sections of the red onion skin, each one cell thick.
- Label 6 watch glasses with the different salt concentrations and place a section of the onion skin in each one for 20 minutes.
- Remove each onion skin section with forceps and place on a slide labeled with the appropriate concentration. Put a drop of the corresponding solution on the slide and float the tissue onto it.
- Cover each section with a coverslip and observe under a microscope soaking up any excess liquid with filter paper.
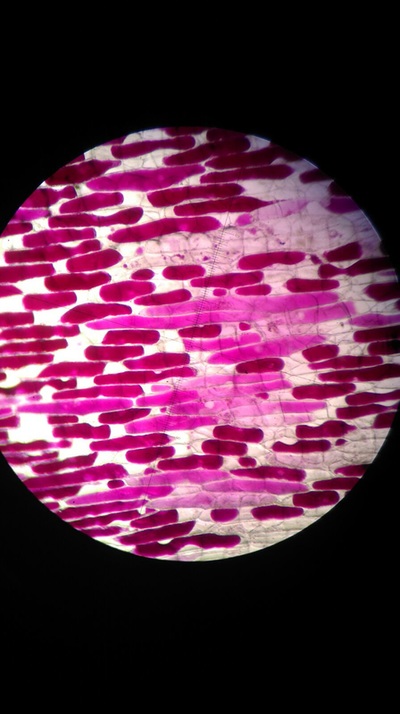
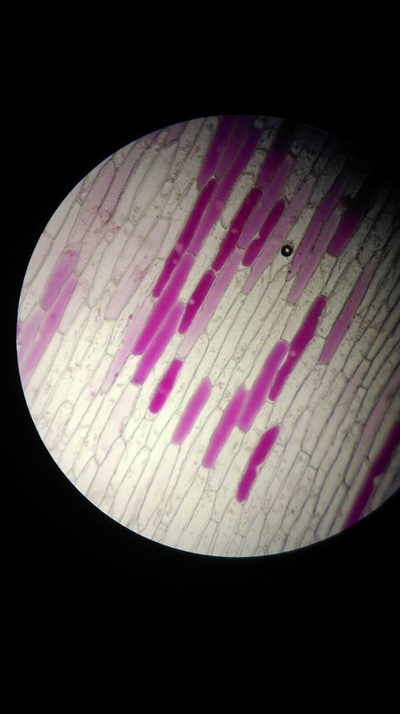
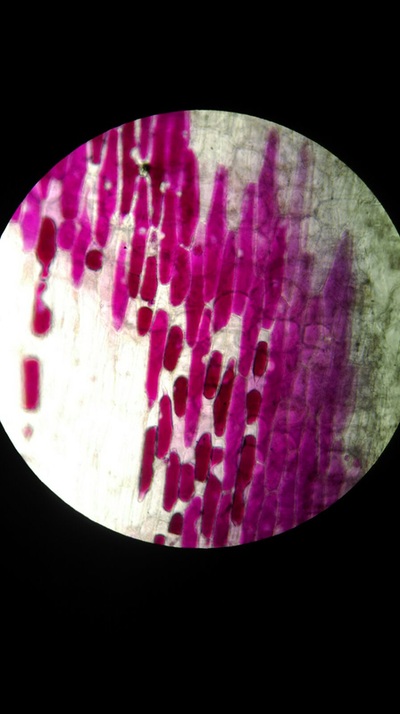
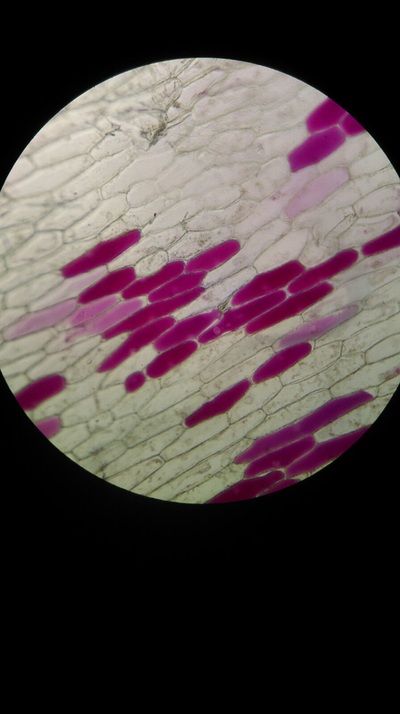
Results:
Conclusion:
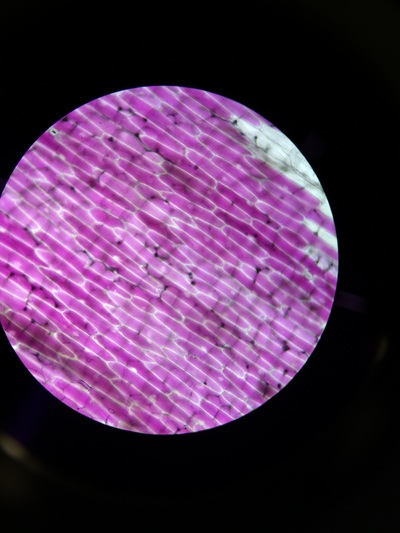
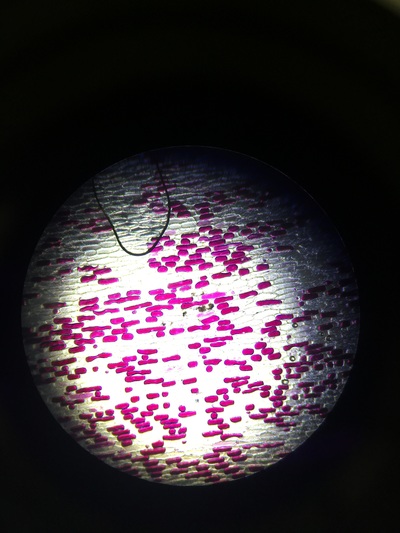
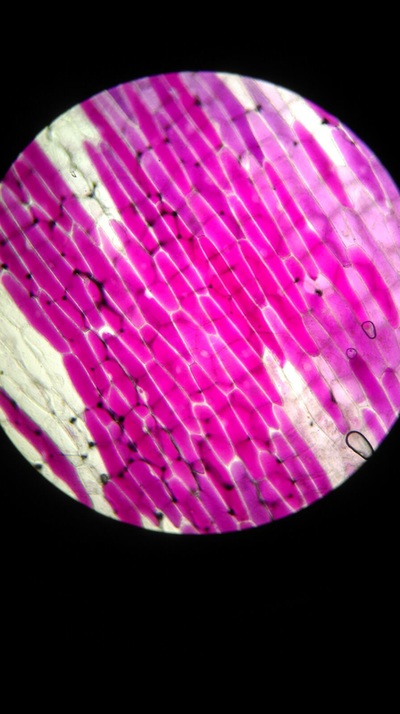
The higher the salt solution concentration the more cells become plasmolysed. This is shown by the 0.1 concentration solution causing no cells to plasmolyse and the 0.6 concentration solution causing 100% of the cells to plasmolyse. The 0.1 solution was isotonic and from 0.2 to 0.6 was hypotonic as water left from the cells into the solution.
The higher the salt solution concentration the more cells become plasmolysed. This is shown by the 0.1 concentration solution causing no cells to plasmolyse and the 0.6 concentration solution causing 100% of the cells to plasmolyse. The 0.1 solution was isotonic and from 0.2 to 0.6 was hypotonic as water left from the cells into the solution.


 RSS Feed
RSS Feed
